by Maritte O’Gallagher
SCIENTIFIC ACHIEVEMENT
Researchers used infrared nanospectroscopy at the Advanced Light Source (ALS) to detect molecular behaviors of ionic liquids—which serve as high performance electrolytes in energy storage devices—under varying charge bias conditions.
SIGNIFICANCE AND IMPACT
Their insights define a direction for targeted design of ionic liquid-based electrolytes with optimized properties for energy storage applications.
An understudied ingredient for high-performance energy storage
Electric double layer capacitors (EDLCs) provide a foundation for a more-resilient energy infrastructure by bridging the performance gap between conventional capacitors and rechargeable batteries. Like batteries, these supercapacitors use a chemical solution called an electrolyte to facilitate the flow of electrical charge between positively and negatively charged electrode surfaces. In EDLCs, two layers of charged particles form where the electrolyte contacts the electrode’s conductive metal surface: charge carriers on the biased electrode and ions in the electrolyte.
Developing advanced electrolytes with optimized properties, such as improved stability under real-world operating conditions or boosted capacity, could enhance the overall performance of these systems. Ionic liquids, which are salts that exist in liquid states under ambient conditions, show promise in their performance as electrolyte components with high electrochemical stability. Still, they are not widely used commercially due to a limited understanding of how ionic liquids behave at electrode surfaces. This is in part because few techniques are capable of detecting how ionic liquids function at the nanoscale level.
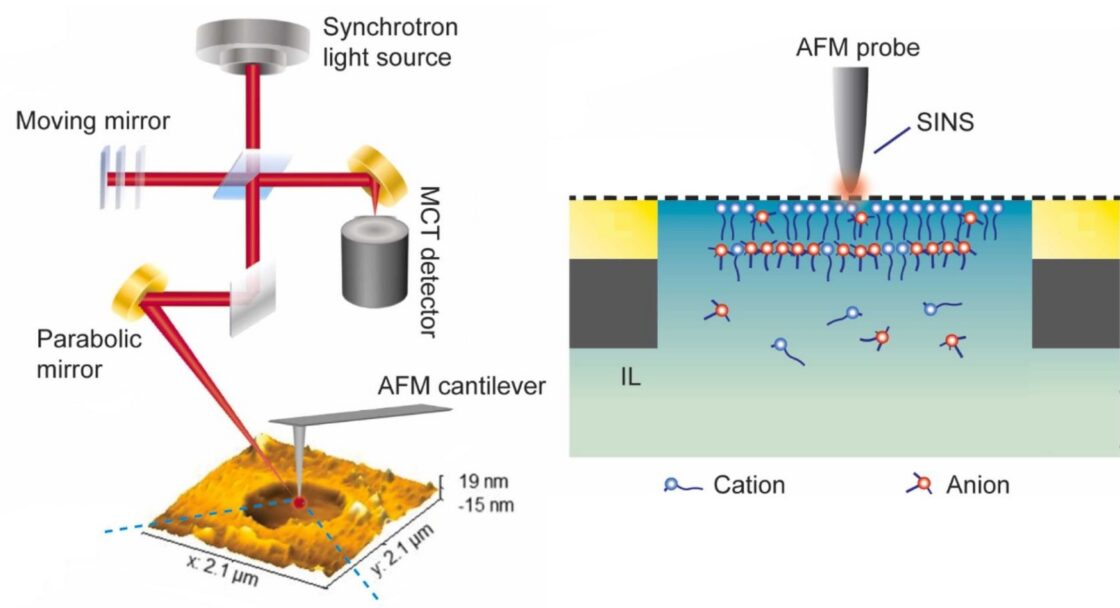
In this work, researchers used infrared nanospectroscopy at the ALS to probe the orientational changes and behaviors of ionic liquids under different applied electrical bias conditions.
Spectroscopy and surface probing uncover unseen electrochemical dynamics
The authors studied the behavior of three different ionic liquids using a specialized non-destructive approach known as scattering-scanning near-field optical microscopy (s-SNOM) which contains an atomic force microscope (AFM) at its core. The AFM produces high-resolution topological maps of material surfaces. s-SNOM involves directing a light source at the AFM tip to probe the chemical and structural properties of the sample on the nanoscale.
While most facilities use a laser for s-SNOM, the ALS infrared beamline used in this study (Beamline 5.4) leverages the synchrotron, which is a more stable light source and provides ultrabroadband coverage of the infrared spectral range. This combination technique, called synchrotron infrared nanospectroscopy (SINS), produces well-defined spectral signatures that correspond to the distinctive stretching and bending of different molecular bonds. These molecular fingerprints reveal which molecules are present and how they are oriented. Studying ionic liquids with SINS had not been done before, and allowed the team to piece together a comprehensive picture correlating the structure and electrochemical properties of ionic liquid electrolytes.
Tailoring ionic liquids for advanced functional applications
SINS data revealed the orientational changes of ionic liquids near electrodes under applied bias. Furthermore, variations in ion size and structure tuned the ionic liquid’s ability to form electric double layer structures with compact ion arrangements, a critical property for enhancing the capacitive performance. The team then correlated these nanoscale electrochemical dynamics with capacitive performance to uncover the underlying principles governing the functional properties of ionic liquids.
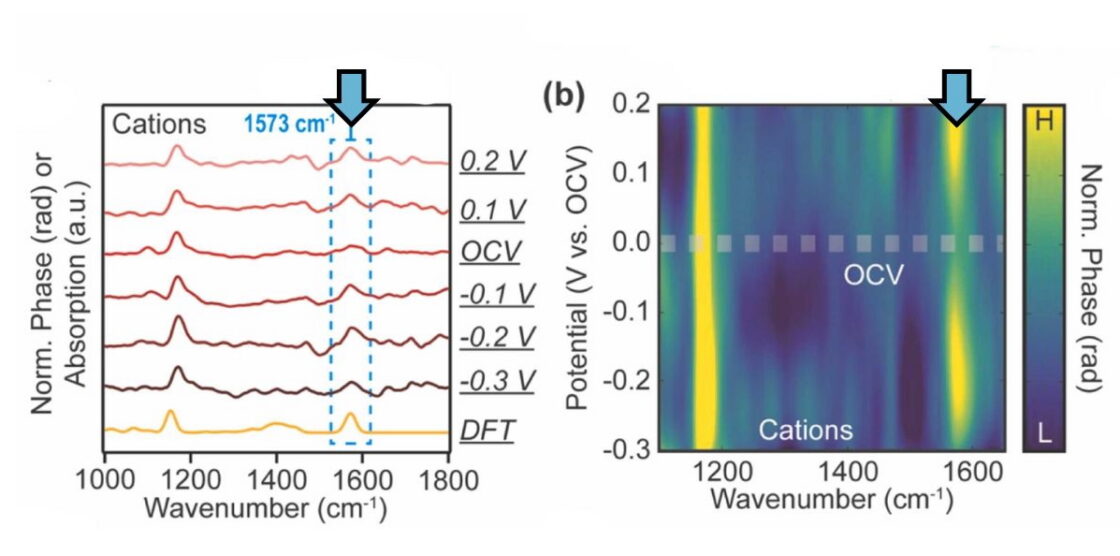
Taken together, findings from this study elucidate how ionic liquids behave at electrode surfaces, representing a significant step towards optimizing the use of ionic liquids in functional applications. They highlight the intricate interactions between ions, and provide insight on how factors like structure and ion size impact capacitor functionality.
Notably, one of the ionic liquids outperformed the other two in terms of its capacitive performance. A larger-scale study could help determine which ionic liquids would be the most suitable for specific electrochemical devices, with different use cases lending specific optimization criteria.
Contacts: Zixuan Li and Stephanie Gilbert Corder
Researchers: Z. Li (ALS and Hunter College of the City University of New York); K.C. Ng and M. Jaugstetter (Berkeley Lab); S. Anderson (University of Wisconsin, Madison); M. Salmeron (Berkeley Lab and University of California, Berkeley); and M.C. Martin, H.A. Bechtel, and S.N. Gilbert Corder (ALS).
Funding: Laboratory Directed Research and Development Program of Lawrence Berkeley National Laboratory. Operation of the ALS is supported by the US Department of Energy, Office of Science, Basic Energy Sciences program (DOE BES).
Publication: Z. Li, K.C. Ng, S. Anderson, M. Jaugstetter, M. Salmeron, M.C. Martin, H.A. Bechtel, and S.N. Gilbert-Corder, “Electrochemical dynamics of imidazolium ionic liquids at graphene electrodes for energy storage applications,” Nano Energy 151, 111785 (2026), doi:10.1016/j.nanoen.2026.111785.
ALS SCIENCE HIGHLIGHT #540