by Brooke Kuei
Joint pain, stiffness, and reduced range of motion are common symptoms of arthritis, a condition that affects half a billion people worldwide. The most widespread form, osteoarthritis, is typically attributed to the gradual breakdown of joint cartilage—but increasing evidence suggests that the inception of this condition could be more complex.
Researchers from the University of California San Francisco (UCSF), the Advanced Light Source (ALS), and the Stanford Synchrotron Radiation Facility (SSRL) recently discovered that subtle mineral changes inside joint tissues may drive early stages of osteoarthritis. In a study published in the Journal of Dental Research, the team combined several x-ray and microscopy techniques to identify chemical elements in joint tissue and how they contribute to alterations in bone mineral structure.
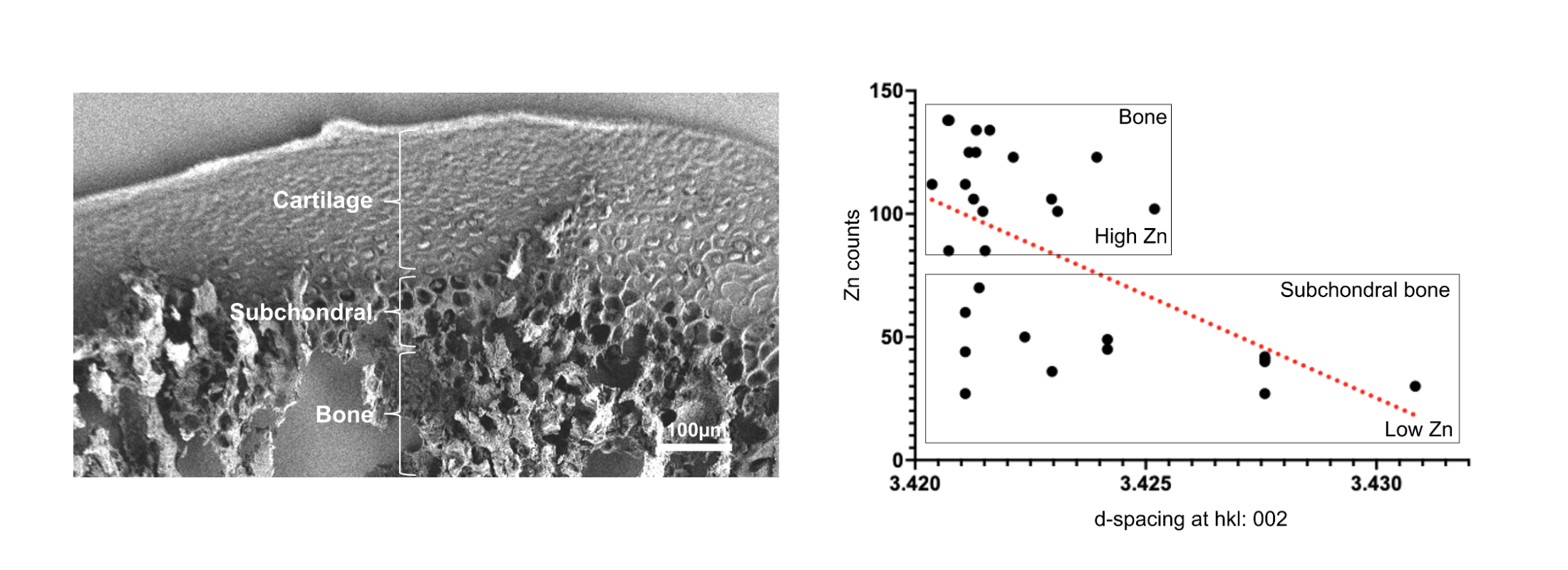
The researchers examined rat temporomandibular joint (TMJ) tissue, informed by human arthritic samples to guide regions of interest, to directly link elemental distribution with changes in mineral crystallinity. They performed x-ray microfluorescence and micro-x-ray absorption near-edge spectroscopy at SSRL to create elemental maps and identify sulfur species in the joint tissue. At ALS Beamline 12.3.2, they used x-ray microfluorescence and x-ray microdiffraction to map where zinc—which is known to be enriched in mineralizing tissue—accumulates in joints and determine how it alters the bone crystallinity.
“Most people think arthritis is just wear and tear,” said Sunita Ho, professor at UCSF’s School of Dentistry. “But we found that tiny mineral changes in joints may quietly push the disease forward.”
At the ALS, x-ray microfluorescence of thin slices of rat TMJ tissue was used to map where zinc was concentrated, while x-ray microdiffraction was used to measure the crystallinity of the bone’s primary mineral component, hydroxylapatite. Beamline 12.3.2’s high spatial resolution allowed the team to examine distinct anatomical regions—located just microns from each other—revealing that transitional regions beneath the cartilage have smaller or less-ordered hydroxylapatite mineral crystals compared to mature bone. The correlative elemental mapping and diffraction studies also revealed that regions with higher zinc levels showed a reduced distance between atomic planes, suggesting that zinc was incorporated directly into the mineral structure.
“The most important aspect of the ALS data is that it connected chemistry to structure,” said Nobumichi Tamura, senior scientist at the ALS. “Other techniques could show that zinc was present, but the microdiffraction experiments explained what zinc was doing to the physical structure of the bone mineral.”
“Right under the cartilage, zinc is integrated into the hydroxylapatite lattice, altering the material properties of subsurface layers between cartilage and bone,” added Ho.
By revealing structural mineral changes that may occur before severe joint damage, this interdisciplinary collaboration between bioengineers and beamline experts opens the door to identifying earlier markers of arthritis. With continued research, such insights could help clinicians detect the disease sooner or reverse its progression.
B.H. Lee, Z. Yang, Y. Wang, J. Levy, N. Tamura, S. Webb, S. Bone, and S.P. Ho, “Sulfur Species in Zinc-Rich Condylar Zones of a Rat Temporomandibular Joint,” J. Dent. Res. (2025), doi:10.1177/00220345251361124.