As climate change raises ocean temperatures, fisheries and public health agencies closely monitor the waters for harmful algal blooms known as red tides. The algae in these blooms produce a neurotoxin that accumulates in shellfish, rendering them dangerous, or even lethal, for human consumption. Bullfrogs, however, have a natural defense in the form of a protein known as saxiphilin.
At the Advanced Light Source (ALS), researchers used high-resolution crystallography at Beamline 8.3.1 to provide structural insights into saxiphilin’s protection mechanism. By studying the protein’s structure with and without the neurotoxin, they found complementary charge-based interactions that explain the high affinity of the neurotoxin for a binding pocket in saxiphilin. Thus, saxiphlin acts as a “molecular sponge,” drawing the neurotoxin away from its intended target, a protein called NaV, and sequestering it. By preventing the toxin from binding to NaV, its paralytic effects are avoided.
Surprisingly, the unrelated family of NaV proteins binds to the neurotoxin through interactions that are similar to how saxiphilin protects the bullfrog. The structural information revealed that neurotoxin recognition evolved convergently in two unrelated proteins, one to deadly effect, and one as a defense.
Most protein targets of toxins studied to date mutate to avoid binding to the attacking toxin. These findings are the first structural characterization of a different defense mechanism—toxin sequestration. The remarkable similarities in how saxiphilin and NaVs recognize a neurotoxin and these newly uncovered structural principles may enable better ways to monitor and combat toxicity levels in the warming oceans and our food supplies.
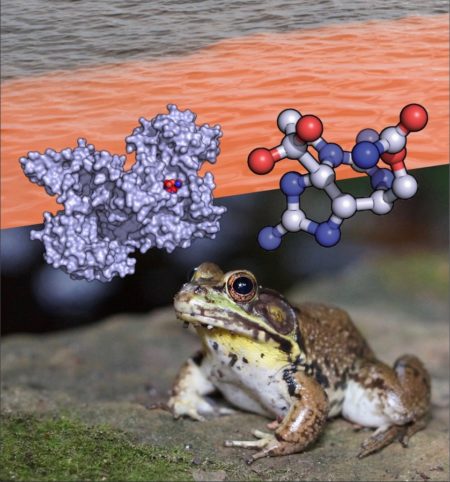
T.-J. Yen, M. Lolicato, R. Thomas-Tran, J. Du Bois, D. L. Minor Jr., “Structure of the saxiphilin:saxitoxin (STX) complex reveals a convergent molecular recognition strategy for paralytic toxins,” Sci. Adv. 5, 6 (2019), doi:10.1126/sciadv.aax2650
See also: A Frog Worth Kissing: Natural Defense Against Red Tide Toxin Found in Bullfrogs