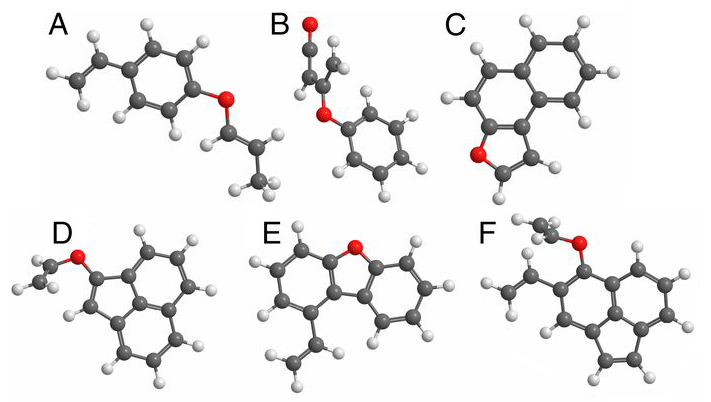
Oxygenated hydrocarbons produced during combustion can have a wide range of detrimental effects on human health, air quality, and regional and global climate. Furans, for example, are compounds that contain rings of four carbon atoms and one oxygen atom. They are frequently observed in the exhaust plumes of combustion sources and nearby environments. Many studies have shown that they are toxic, whether ingested or inhaled, and thus pose a considerable threat to human health.
Despite several studies, however, their precise chemical formation pathways from combustion sources and their link to carcinogenic polycyclic aromatic hydrocarbon (PAH) molecules have remained unknown. In this study, researchers report for the first time the formation of large oxygenated hydrocarbons during the combustion of very simple fuels, such as ethylene. The results demonstrate that furans are present in soot from the beginning and are incorporated into particles as they grow.
The researchers used first-principles electronic-structure calculations in conjunction with probabilistic models to study the formation and growth of the particles using atomistic simulations. The resulting predictions were validated in well-characterized test flames, using vacuum-ultraviolet (VUV) photoionization aerosol mass spectrometry at ALS Beamline 9.0.2, which allowed identification of oxygenated species by mass. X-ray photoelectron spectroscopy measurements (using a laboratory x-ray source) identified the oxygen functional groups present in particles.
The study advances our understanding of the oxidation chemistry of organic carbon, which is critical to many processes, from controlling emissions of toxic combustion by-products to reducing anthropogenic climate change.
Work performed at ALS Beamline 9.0.
K.O. Johansson, T. Dillstrom, M. Monti, F. El Gabaly, M.F. Campbell, P.E. Schrader, D.M. Popolan-Vaida, N.K. Richards-Henderson, K.R. Wilson, A. Violi, and H.A. Michelsen, “Formation and emission of large furans and other oxygenated hydrocarbons from flames,” Proc. Natl. Acad. Sci. USA 113, 8374 (2016).