by Gianna FazioLiu
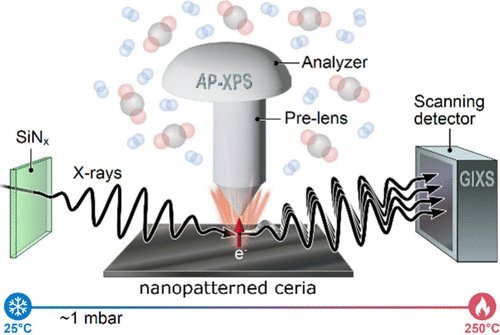
Ceria (cerium oxide) is particularly valued for its ability to store, release, and shuttle hydrogen under reaction conditions. However, understanding how ceria simultaneously undergoes chemical and structural transformations during catalysis has remained a persistent challenge, as these changes evolve dynamically under reactive environments.
In a recent study at the Advanced Light Source (ALS), researchers employed an approach that uses a single X-ray beam to capture both chemical and structural changes in nanopatterned ceria during catalytic reactions.
By combining ambient-pressure x-ray photoelectron spectroscopy (APXPS) with grazing-incidence x-ray scattering (GIXS), the team was able to observe how ceria interacts with hydrogen and carbon dioxide in real time. This combined method, termed ambient-pressure photoemission and X-ray scattering (APPEXS), provided a dual-mode measurement. This measurement revealed a hierarchy of electron density changes associated with hydrogen uptake, providing new insight into the mechanisms underlying ceria’s catalytic performance. Additionally, the ability to identify roughness changes and oxidation state changes were key outcomes.
“Our goal was to access both surface and bulk data simultaneously through capturing chemistry and structure in a single measurement,” explained Baran Eren, senior scientist in the Faculty of Chemistry at Weizmann Institute of Science. “Rather than treating structure and chemistry as separate observables, we can now see how materials behave together under realistic reaction conditions.”
Currently available only at ALS Beamline 11.0.2.1, this capability was developed by Slavo Nemsak. His pioneering approach, which earned him the 2023 Klaus Halbach Instrumentation Award, has since inspired similar efforts at other synchrotron facilities across Europe and Asia.
The researchers examined nanopatterned ceria exposed to hydrogen and oxidizing environments. They found that hydrogen incorporation—resulting in the formation of hydride and oxyhydroxide species—drives significant restructuring of the material’s surface, especially when oxygen is present from air or carbon dioxide. As ceria absorbs hydrogen to facilitate reactions, its interior becomes denser while its surface roughens, reflecting a coupled shift in both oxidation state and physical morphology. These findings indicate that defect-rich microstructures play a key role in enabling subsurface hydrogen diffusion at lower temperatures than previously anticipated.
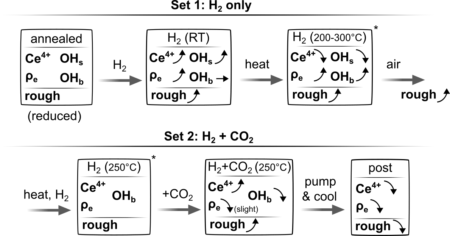
Because these chemical and structural transformations occur in tandem, the study underscores the importance of simultaneous measurement techniques for accurately characterizing and optimizing catalytic materials. The results provide critical guidance for engineers seeking to design more-efficient ceria-based catalysts, particularly for applications where hydrogen activation and transport are central to performance.
The next steps will extend this approach to size-controlled nanoparticles and, ultimately, to industrial catalysts, where complexity is significantly higher but also more relevant for real-world applications. Looking ahead, the methodology could also be applied to electrochemical systems, opening new opportunities to probe catalytic energy-relevant processes under realistic operating conditions.
A. Ben Yaacov, M. Jaugstetter, H. Kersell, O. S. Bitton, M. B. Salmeron, S. Nemšák, and B. Eren, “One Beam, Dual Insights: Simultaneous Chemical and Structural Changes in Nanopatterned Ceria under Reaction Conditions,” J. Phys. Chem. Lett. (2026), doi:10.1021/acs.jpclett.6c00039