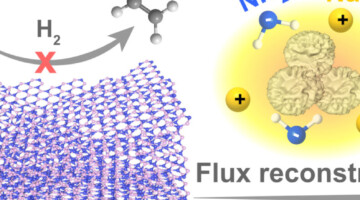
Researchers engineered defects in boron nitride to develop a metal-free route for purifying ethylene to remove acetylene. The ALS shed light on how the engineered defects were responsible for exceptional selectivity. Read more »
Reaction Mechanism of Commercial Lithium-Ion Battery Cathodes
Researchers used soft x-ray resonant inelastic x-ray scattering at the ALS to understand the role of aluminum doping in improving the stability of commercially used cathode materials for lithium-ion batteries. Read more »
Mind-Blowing Materials: Mimicking Neurons for Faster Computing
Researchers used x-ray absorption spectroscopy and resonant inelastic x-ray scattering at the ALS to uncover the atomic-level mechanism of conductance switching for a neuromorphic material that has the potential for energy-efficient computing. Read more »
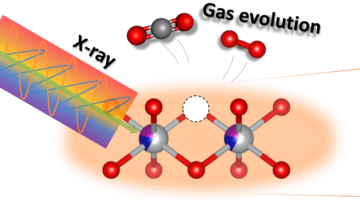
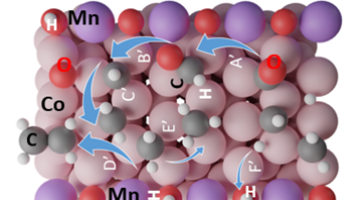
Understanding the Role of Manganese in Fuel Production Catalysts
Using specialized equipment at the Advanced Light Source (ALS), including a custom-built reaction cell, researchers uncovered the role of manganese in cobalt manganese oxide catalysts used for fuel production. Read more »![]()
![]()
Manganese Cathodes Could Boost Lithium-ion Batteries
Rechargeable lithium-ion batteries are used in mobile devices, electric vehicles, and energy storage systems. But supplies of nickel and cobalt, commonly used in the cathodes of these batteries, are limited. New research opens up a potential low-cost, safe alternative in manganese, the fifth most abundant metal in the Earth’s crust. Read more »
Cobalt or Nickel: Which is Better for High-Energy Battery Cathodes?
High-energy Li-ion batteries can provide both high capacity and high voltage, both of which are important in electric vehicles for greater range and faster acceleration. Here, researchers untangled the contributions of nickel and cobalt in high-energy Li-rich battery cathodes, pointing the way to optimizing them via a compositional approach. Read more »
HyMARC Aims to Hit Targets for Hydrogen Storage Using X-Ray Science
Understanding how materials absorb and release hydrogen is the focus of the Hydrogen Materials Advanced Research Consortium (HyMARC). At the ALS, the HyMARC Approved Program was recently renewed, underscoring the key role that soft x-ray techniques have played in addressing the challenges of hydrogen storage. Read more »
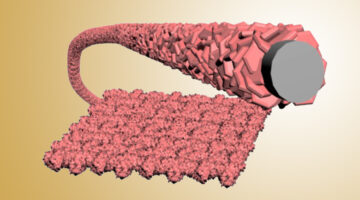
Surface Engineering Boosts Water-Splitting Efficiency
Researchers modified the surface of an electrocatalyst to maximize its efficiency at splitting water. The optimized material is approximately 40 times more efficient than similar commercial electrocatalysts and could help make the production of clean hydrogen fuel more sustainable and economical. Read more »![]()
![]()
Increasing the Energy Density of Hybrid Supercapacitor Electrodes
Hybrid supercapacitors (HSCs) integrate the merits of batteries with those of supercapacitors. However, the fraction of active material in HSC electrodes has remained too low for commercial requirements. Now, researchers have found a clever way to increase the active-mass ratio to achieve dramatic improvements in key measures. Read more »
Electric Vehicle Batteries Could Get Big Boost With New Polymer Coating
Scientists have developed a conductive polymer coating—called HOS-PFM—that conducts both electrons and ions at the same time. This ensures battery stability and high charge/discharge rates while enhancing battery life. The coating also shows promise as a battery adhesive that could extend the lifetime of a lithium-ion battery from an average of 10 years to about 15 years. Read more »
- 1
- 2
- 3
- …
- 5
- Next Page »