In shifting from fossil fuels to renewable energy, a promising strategy is to convert the greenhouse gas, carbon dioxide (CO2), into useful liquid fuels and chemicals. Since copper (Cu) remains the sole element able to catalyze the reduction of CO2 to valuable products, significant effort has been devoted to developing Cu electrocatalysts with improved selectivity and activity. While information about Cu chemical states and particle structure under reaction conditions would help in these efforts, such “operando” experiments had previously been unfeasible, particularly for high-performance Cu nanocatalysts.
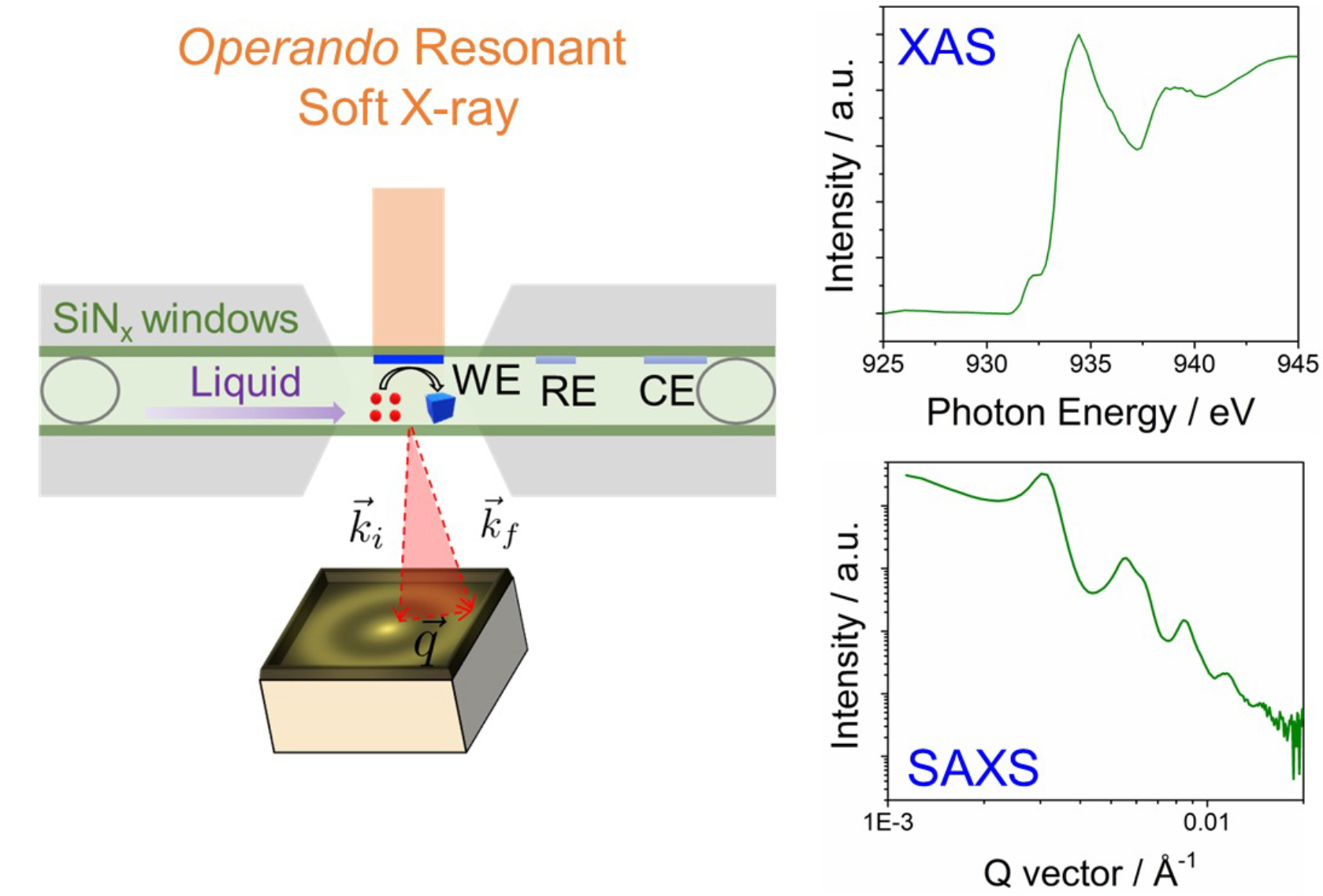
Now, at Advanced Light Source (ALS) Beamline 11.0.1.2, researchers have developed an operando resonant soft x-ray scattering (RSoXS) capability that enables correlated x-ray absorption spectroscopy (XAS) and small-angle x-ray scattering (SAXS) through a 1 µm thick liquid electrolyte layer. The method is surface sensitive and probes the valence-electron transition directly relevant to the catalytic center of Cu active sites. In addition, SAXS offers a statistical analysis of particle-to-particle interactions in nanoparticle ensembles.
In this work, the operando XAS technique was used to investigate the stability of Cu nanocatalysts exposed to the x-ray beam. It also probed the nanoparticles’ voltage-dependent structural evolution. The high sensitivity of scattering-enhanced XAS allows the detection of chemical valence with subsecond x-ray pulses.
Small nanoparticles (7 nm) showed irreversible soft-x-ray-induced beam oxidation of Cu to CuO in liquid when exposed to x-rays for only half a second. In comparison, large nanoparticles (18 nm) showed improved stability, enabling the reliable electroreduction of surface Cu2O to metallic Cu.
“This is the first operando RSoXS study on electrochemical systems,” said Cheng Wang, ALS beamline scientist and co-author of the study. “It’s a technical breakthrough that will open up RSoXS to the electrochemistry community.”
The study also points out the need for rigorous examination of beam effects (x-ray or electron) when performing operando x-ray studies on electrocatalysts in general, and beam-sensitive Cu catalysts for CO2 reduction in particular. The strategy of correlating x-ray spectroscopy and small-angle x-ray scattering can serve as a powerful analytical technique for simultaneously probing chemical valence and interparticle dynamics for a variety of energy-related electrochemical reactions under realistic operating conditions.
Y. Yang, I. Roh, S. Louisia, C. Chen, J. Jin, S. Yu, M.B. Salmeron, C. Wang, and P. Yang, “Operando Resonant Soft X-ray Scattering Studies of Chemical Environment and Interparticle Dynamics of Cu Nanocatalysts for CO2 Electroreduction,” J. Am. Chem. Soc. 144, 8927 (2022), doi:10.1021/jacs.2c03662.