by Maritte O’Gallagher
SCIENTIFIC ACHIEVEMENT
Researchers identified a compound that disrupts a hard-to-target tumor growth pathway in breast, lung, and other cancers and used the Advanced Light Source (ALS) to characterize the chemical interactions critical to its potency.
SIGNIFICANCE AND IMPACT
This work contributed to the development of a similar compound currently undergoing clinical trials in cancer patients, and informs hypotheses for designing better drug candidates.

The cancer growth pathway once called undruggable
RAS genes are a family of genes involved in regulating cell growth and division. Mutations in these genes drive over 22% of all human cancers by acting like broken molecular switches stuck in the on position, constantly activating multiple downstream signaling pathways that trigger uncontrolled cell proliferation.
Treating these aggressive cancers requires a multipronged therapeutic approach that targets several RAS-activated signaling pathways. While phosphoinositide 3-kinase alpha (PI3Kα) is one of these key pathways, PI3Kα also interacts with insulin to control sugar levels. So cancer patients treated with the PI3Kα inhibitors that are currently available often experience debilitating high blood glucose symptoms, reducing their ability to take effective doses of the medicine for long periods.
Prior studies have shown that genetically disrupting the interaction between RAS and PI3Kα limits RAS-driven tumor growth without the negative impacts on insulin levels seen with traditional PI3Kα inhibitors. While existing PI3Kα inhibitors affect the more general activity of PI3Kα, targeting the protein–protein interaction of RAS and PI3Kα appears to selectively disrupt cancer-related signalling while leaving normal cellular activities unscathed. Researchers at San Diego-based biotechnology company Vividion Therapeutics recognized this as a potential opportunity to selectively target cancerous cells while sparing healthy cells. They identified a candidate compound that binds stably and selectively to the RAS-binding region of PI3Kα and characterized how the new molecule interacted with PI3Kα using x-ray diffraction studies at the ALS.
Crystallography reveals nuances of PI3Kα inhibition
The researchers crystallized a small piece of the PI3Kα protein bound to their newly identified inhibitor and sent crystals of the complex to the ALS. The x-ray diffraction data collected at Beamline 5.0.2 enabled them to resolve the molecular structure of the complex, which helped uncover the mechanism by which the inhibitor disrupts the RAS–PI3Kα interaction. Rather than sitting in the spot where RAS would bind, Vividion’s compound prevented the PI3Kα protein from adopting the right pose to interact with RAS.
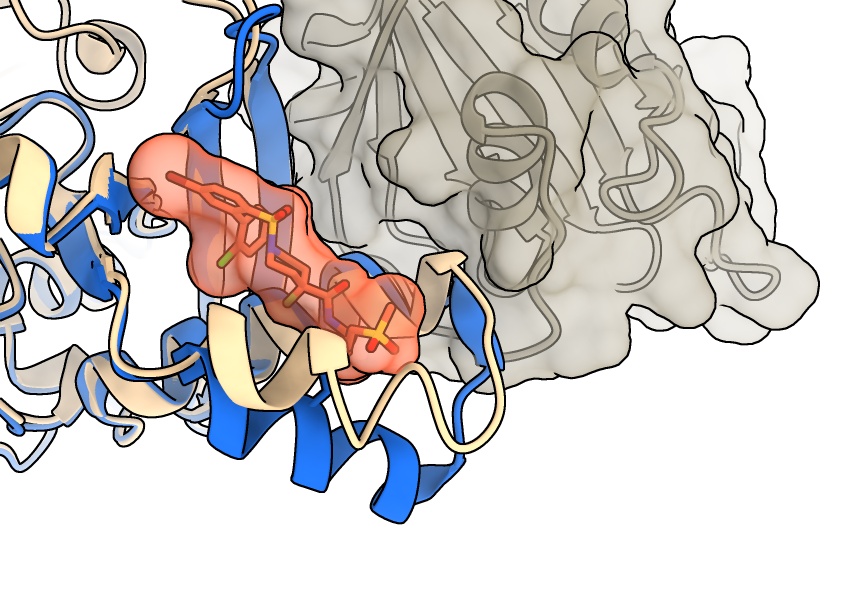
Additional laboratory experiments bolstered these results, confirming that the new inhibitor blocked PI3Kα activity and tumor growth in a wide range of cancer models without disrupting normal cellular activities. This safety profile indicates a strong potential for the strategy to be used in combination with other therapies targeting the RAS pathway, which is supported by preclinical models showing enhanced efficacy of combination treatments.
A new strategy in the fight against RAS-driven cancers
Due to the hard-to-treat, aggressive nature of RAS-driven cancers, a therapeutic toolbelt composed of many different approaches is necessary to effectively combat them. The discovery of molecules that selectively disrupt the RAS–PI3Kα interaction represents a new tool in the battle against these cancers.
Long term, this strategy could provide meaningful therapeutic benefit to an underserved patient population, fighting tumors without decreasing patients’ quality of life. The work presented here directly contributed to the development of a similar, further optimized compound currently in a clinical trial evaluating its utility in treating patients with RAS-driven and related cancer types.
The Vividion team continues to pursue new cancer therapies using similar structure-based drug design strategies, fueled by data collected at the ALS and other DOE-funded synchrotrons.
Additional Press:
Vividion Therapeutics
The Crick Institute
Contacts: Steffen Bernard
Researchers: J.E. Klebba, N. Roy, S.M. Bernard, S. Grabow, M.A. Hoffman, H. Miao, J. Tamiya, J. Wang, C. Berry, A. Esparza-Oros, R. Lin, Y. Liu, M. Pariollaud, H. Parker, I. Mochalkin, A.N. Snead, E.J. Walton, T.E. Wyrick, E. Aitichson, K. Bedke, B.D. Horning, K.N. Lamb, W. Lin, J. Lu, M.K. Pastuszka, J. Pollock, J.J. Sigler, C. Yue, B.N. Cook, G.M. Simon, D.S. Weinstein, and M.P. Patricelli (Vividion Therapeutics); S. Rana, M. Ismail, M. Tomaschko, M. Molina-Arcas, and J. Downward (Francis Crick Institute); J.C. Brannon (Odyssey Therapeutics); J.M. Chick and T.M. Kinsella (Independent researchers); K. Hee (SciexScie); and E. Tran (Novartis Institutes for Biomedical Research).
Funding: Cancer Research UK, UK Medical Research Council, Wellcome Trust, European Research Council Advanced Grant RASImmune, Howard Hughes Medical Institute, National Institutes of Health (National Institute of General Medical Sciences). Operation of the ALS is supported by the US Department of Energy, Office of Science, Basic Energy Sciences program (DOE BES).
Publication: J.E. Klebba, N. Roy, S.M. Bernard, S. Grabow, M.A. Hoffman, H. Miao, J. Tamiya, J. Wang, C. Berry, A. Esparza-Oros, R. Lin, Y. Liu, M. Pariollaud, H. Parker, I. Mochalkin, S. Rana, A.N. Snead, E.J. Walton, T.E. Wyrick, E. Aitichson, K. Bedke, J.C. Brannon, J.M. Chick, K. Hee, B.D. Horning, M. Ismail, K.N. Lamb, W. Lin, J. Lu, M.K. Pastuszka, J. Pollock, J.J. Sigler, M. Tomaschko, E. Tran, C. Yue, T.M. Kinsella, M. Molina-Arcas, B.N. Cook, G.M. Simon, D.S. Weinstein, J. Downward, and M.P. Patricelli, “Covalent inhibitors of the PI3Kα RAS binding domain impair tumor growth driven by RAS and HER2,” Science 390, 702 (2025), doi:10.1126/science.adv2684.
ALS SCIENCE HIGHLIGHT #536