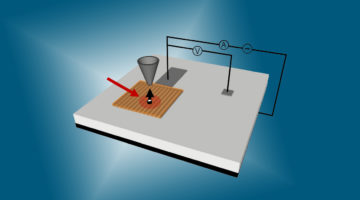
The rate-limiting step in catalysis involving oxygen uptake was identified through analysis of the reaction pathways and observations performed under operating conditions. The work lays the foundation for improving the efficiency of energy conversion and storage devices such as fuel cells, catalytic reactors, and batteries. Read more »![]()
![]()
New Catalyst Resists Destructive Carbon Buildup in Electrodes
Key challenges in the transition to sustainable energy can be met by converting CO2 to CO through the use of solid oxide electrolysis cells. But because these can suffer from carbon deposition at the electrodes, researchers have now identified and tested a new, cerium oxide–based catalyst that is more resistant to carbon buildup. Read more »![]()
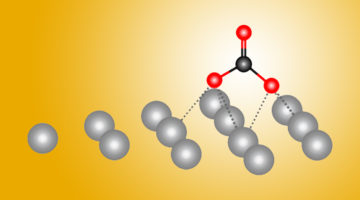
A New Path to Carbon Dioxide Transformation
Combining ALS experiments with quantum-mechanical calculations, scientists found dramatic differences in how carbon dioxide (CO2) reactions begin on silver as opposed to copper. Both metals help transform CO2—a greenhouse gas—into more useful forms, and this new atomic-level data could help make the process more efficient. Read more »![]()
![]()
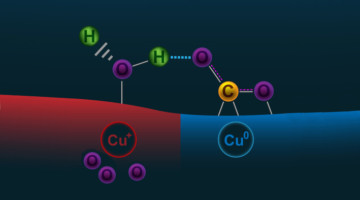
Subsurface Oxygen Boosts Activity of Copper Catalysts
Scientists are seeking ways to reduce levels of CO2 in the atmosphere by improving the processes that convert CO2 gas into ethanol (a liquid fuel). But copper, the best catalyst for this, is not very efficient. Now, ambient-pressure x-ray experiments have revealed how subsurface oxygen boosts copper’s catalytic activity. Read more »![]()
![]()
Researchers Find a Surprise Just Beneath the Surface in Carbon Dioxide Experiment
X-ray experiments, coupled with theoretical work, revealed how oxygen atoms embedded near the surface of a copper sample had a more dramatic effect on the early stages of a reaction with CO2 than accounted for in earlier theories. This work could prove useful in designing new catalysts for converting CO2 into liquid fuels and other products. Read more »
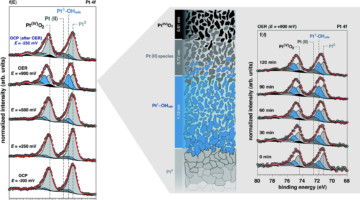
A Closer Look at a Working Platinum/Electrolyte Interface
Ambient-pressure studies of the interface between a platinum electrode and an alkaline electrolyte revealed the molecular-level chemistry, structure, and dynamics of the platinum surface as a function of applied potential, highlighting differences between thermodynamic predictions and the actual surface composition. Read more »
A Surface Treatment for Improving Fuel-Cell Cathodes
Solid-oxide fuel cells (SOFCs) are a promising path toward the “clean” conversion of chemical energy to electrical energy with little or no carbon dioxide emission. With the help of the ALS, researchers from MIT recently found a way to treat SOFC cathode surfaces so that they perform better and last longer. Read more »![]()
![]()
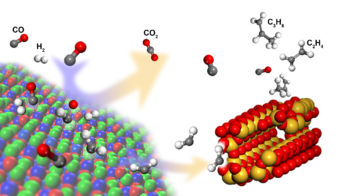
New Catalyst Boosts Selective Formation of Olefins from Syngas
Experiments at the ALS have helped to explain how a new catalyst significantly boosts the selective formation of light olefin molecules—important building blocks in the petrochemical industry—from syngas. The new process could allow for the use of alternative syngas feedstocks that save water and energy. Read more »![]()
![]()
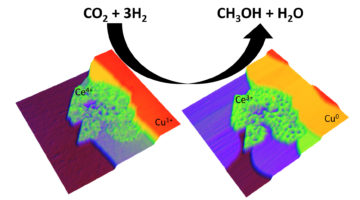
From CO2 to Methanol via Novel Nanocatalysts
Researchers have found novel nanocatalysts that lower the barrier to converting carbon dioxide—an abundant greenhouse gas—into methanol—a key commodity used to produce numerous industrial chemicals and fuels. In one case, it worked almost 90 times faster than catalysts commonly used for this reaction today. Read more »![]()
![]()
Platinum Nanoclusters Out-Perform Single Crystals
Researchers have found that under high pressure—comparable to the pressures at which many industrial technologies operate—platinum surfaces can change their structure dramatically in response to the presence of high-coverage reactants. Read more »![]()
![]()